Stem Cell Therapy Face

Stem Cell Therapy Face
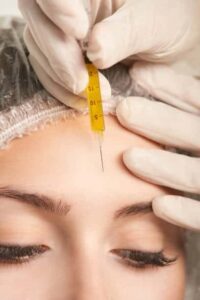
Stem cell therapy is a completely natural, injection based treatment which is designed to stimulate the body’s regenerative abilities. Prior to treatment, a stem cell banks living stem cells are harvested and concentrated into an injectable serum. In many cases other blood cells or cell-like particles, i.e. Hyaloronic acid, exosomes or platelets, are included into the mix. Once introduced into damaged tissue a powerful, localized healing response results. In this way chronically damaged structures can be made to heal properly and completely.
Call Today 724-683-7581
It’s an innovative treatment that’s becoming increasingly popular on its own. Yet when combined with PRP it’s even more effective. Prior to microneedling, several vials of a patient’s own blood are drawn. Using a laboratory centrifuge this blood is separated into its constituent layers. The platelet-rich portion is drawn off and further processed into a solution which can further stimulate the healing process. Right now PRP therapy is used almost exclusively to treat stubborn, chronic musculoskeletal injuries. When administered alongside microneedling, however, it triggers this same healing response in the skin of the face. It’s a very practical match.
Not surprisingly, the results of the vampire facelift resemble those of microneedling combined with injectable facial fillers. These products, the most popular of which is probably Juvaderm, physically add volume to the face. Once in place they temporarily compensating for age-related changes.
PRP when combined with microneedling prompts the face to pump out more collagen. This leads to filler-like results that are entirely natural and often last many months longer than even the best injectable fillers.
Medical Residency and Fellowship training?
Ideally, the doctor you’re considering should have both. These serve as concrete proof of advanced training and a high degree of experience. Residency programs and Fellowships are intense periods of hands-on learning which take place in real hospitals or clinics with actual patients. They typically last 2, 3, 4, or even 5 years.
These programs are usually highly competitive, and designed to attract the best of the best. When choosing a Regenerative Medicine doctor, check to see if they’ve done a Residency or Fellowship. This will usually be in Orthopedic Medicine or another joint-related specialty. Our Surgeon has a fellowship truly related to stem cell transplantation. After a 5 year General Surgery Residency at AGH, he pursued a 3 year Transplant Surgery at UPMC. He then accomplished an additional fellowship with a renowned plastic surgeon and became Board Certified in Cosmetic Surgery.
Professional Societies

- American Academy of Cosmetic Surgery
- American Society of Cosmetic Laser Surgery
- American Society of Liposuction Surgery
- American College of Medical Quality
- American Society of Laser Medicine and Surgery
- American Society of Transplant Surgeons
- Harvard Medical School Postgraduate Association
- American Vein and Lymphatic Society
- American Medical Association
There is no physician more uniquely well trained to oversee your treatment. He can even transfer fat cells containing stem cells from your own fatty areas through liposuction, and transfers then to the fat for a nonsurgical facelift.
Are they using Stem Cells from the right source?
Doctors who perform Regenerative Medicine order living Stem Cell cultures from medical supply companies, almost without exception. Yet not all Stem Cell cultures are created equal. There are 2 sources for commercially available Stem Cells—Umbilical and Amniotic. Both are harvested at the time of birth without harming the mother or newborn, then purified into usable cell cultures.
Umbilical Stem Cells are harvested from the tissue of the umbilical cord, which contains very high concentrations of living cells. Stem Cell cultures from Umbilical sources are therefore of the highest quality. Cultures can also be prepared from an Amniotic Fluid source, though. These are significantly less expensive. They’re also of a much lower quality and contain far fewer living, healthy Stem Cells. The choice is clear, unless you’re shopping entirely by price.
This list also applies to choosing any type of doctor in a general sense. Doctors with the right certifications, training, and a strong focus on their specialty will almost always be the best. Choose your providers carefully, and always do your homework before going through with any medical treatment.
Postnatal Stem Cells and FDA Regulations
Platelet-Rich plasma preparation. Plasma in syringe. Centrifuge. Background
Recently, there have been several nationally published articles concerning the use of postnatal stem cells and the governing regulations of the FDA. The FDA has acknowledged that stem cell products have the potential to treat many medical conditions and diseases and have strict regulatory restrictions in place and often review manufacturers processes and procedures for compliance.
As part of the FDA’s review, investigators must show how each product will be manufactured so the FDA can make sure appropriate steps are being taken to help assure the product’s safety, purity, and strength (potency).
We are proud of our 14-year history of remaining compliant without any violations issued.
Through our strict processes, we take every precaution available to ensure that our products are safe, in their purest form and maintain the highest quality of potency.
ELIGIBILTY
Our proprietary process begins with a meticulous screening of the donor mother and father as well as family members for several generations.
COLLECTION
The Director of Procurement will notify the Recovery Specialist that a donation is ready at the designated hospital. The Recovery Specialist will collect the donation and return it directly back to Utah Cord Bank to be processed.
TESTING
Our lots are quarantined for 14 days and thoroughly tested for bacterial and disease contamination by a CLIA certified lab.
PROCESSING
Our proprietary process meets the FDA’s requirements of cells and tissues being minimally manipulated.
Are there any age restrictions for individuals seeking stem cell facial rejuvenation?
There isn’t a strict age restriction for stem cell facial rejuvenation; eligibility depends on the individual’s overall health and specific concerns. Generally, adults seeking facial rejuvenation, especially to address issues like wrinkles, fine lines, or skin sagging, are suitable candidates. The treatment aims to enhance skin quality and rejuvenate appearance, making it appealing to a wide age range. However, the effectiveness can vary based on skin condition and age. Younger individuals may seek preventive measures or address early signs of aging, while older adults may target more advanced concerns. A thorough consultation with a qualified practitioner helps determine suitability and establish realistic expectations based on individual needs.
How long does a typical stem cell facial procedure take, and is it performed as an outpatient treatment?
The duration of a stem cell facial procedure varies based on the specific technique used and the areas being treated. Generally, these procedures are minimally invasive and can typically be completed within a few hours. Stem cell therapy for the face is often performed as an outpatient treatment, allowing patients to go home the same day. Local anesthesia or numbing creams are commonly used to ensure comfort during the procedure. While the treatment time is relatively short, patients should plan for some recovery time afterward, during which they might experience mild swelling or redness at the treatment site.
Can stem cell therapy help with specific skin conditions, such as acne scars or hyperpigmentation?
Yes, stem cell therapy shows promise in addressing specific skin conditions like acne scars and hyperpigmentation. Stem cells have regenerative properties that may promote skin healing, collagen production, and even out skin tone. However, individual responses can vary, and outcomes depend on factors such as the severity of the condition, the specific procedure used, and the patient’s overall health. Consultation with a specialist is essential for personalized advice.
How Long A Stem Cell Therapy Session Usually Takes?
Normally, a stem cell therapy session may take anywhere from 1 to 4 hours, but it depends on the area being treated. But the duration can vary as well, since there will be time needed to prepare stem cells, administrating the injection, or observing the patient after the treatment to make sure there are no side effects afterwards.
Does Stem Cell Therapy Increase The Risk Of Tumors or Cancers?
Yes, unfortunately there are risks of tumors or cancers from stem cell therapy, but the risk will depend on the type of stem cells used. For example, embryonic and induced pluripotent stem cells have much higher risks of uncontrolled growth, which can increase the chances of tumors. If stem cell treatments are improperly regulated then it also increased the chances of cancer, as cells may mutate or fail to differentiate. Luckily, these risks and complications can be easily minimized if you take the treatment from reputable clinics that follow strict safety regulations and conduct through testing before administrating therapy.
What Are The Successful Rates If Stem Cell Treatments For Various Conditions?
The success rates will of stem cell treatments vary depending on the condition that’s being treated, the type of stem cells used, and individual patient factors. If you have orthopedic issues that include joint pain, and osteoarthritis, then the success rate of stem cell therapy on you can be 60-80%, many patients who get this treatment experience pain relief and improved mobility. If you suffer from blood-related diseases like leukemia, then stem cell transplant have a high success rate, 70% at maximum. Unfortunately, for neurodegenerative diseases like Parkinson’s or Alzheimer’s, there isn’t a lot of research done, so the results remain inconsistent. But at least in for most conditions, this treatment has a high success rate!
How Stem Cells Are Obtained For Therapy?
Stem cells can be obtained from multiple sources, depending on the type of treatment you might get. Most adult stem cells are usually harvested from bone marrow, umbilical cord blood and placental tissues also provide rich sources of stem cells, these are then collected at birth for future use. Each source has different properties, with varying potential for regeneration and therapeutic applications.
Does Stem Cell Therapy Have Any Side Effects?
Yes, every type of treatment carries a risk of side effects and it’s possible with stem cell therapy as well. If you are wondering about the side effects, then you should be careful above infection at the injection site, bleeding, inflammation and discomfort, fortunately none of them are serious! Furthermore, if stem cells are not processed properly or are used incorrectly, then there may be risks of unwanted tissue growth, which can include even tumors. There are risks such as rejection or immune reactions too, but all of this is pretty rare so you don’t need to be really worried. But be sure to discuss about allergies or any chronic condition, if you are suffering from any.
Is Stem Cell Therapy Safe For Kids?
Yes, stem cell therapy can be safe for kids, especially when it’s used appropriately and under supervision of qualified medical professionals! However, you need to consider that stem cell therapy is used for specific conditions such as genetic disorders, blood cancers, or congenital diseases. Also, the effectiveness will depend on the condition which is being treated the type of cells used, and the experience of healthcare provider.
Can Stem Cell Therapy Help With Chronic Pain?
Yes, stem cell therapy can easily help out with chronic pain, especially conditions like osteoarthritis, joint injuries, and degenerative disc disease. Stem cell also promotes healing and reduces inflammation to the damaged tissues.
UTAH CORD BANK EMPHASIZES A 100% CHAIN OF CUSTODY
Collection of Postnatal Donation A Recovery Specialist, once given the donation information from the Director of Procurement, will collect the postnatal donation at the designated hospital. The Recovery Specialist will then return the donation to UCB to be processed.
Donor Eligibility Requirement Our proprietary process begins with meticulous screening of the donor mother and father as well as family members for several generations. In addition to routine industry screens for blood borne pathogens, we also screen for heritable and non-heritable conditions, environmental contaminants from medications, alcohol, drugs, tobacco and electronic vaping. Only healthy families who met or exceed these criteria are considered. Postnatal tissues can be collected within 30 minutes of a Cesarean section. After processing, allografts are tested again to ensure they are free of contaminants or infection, and contain healthy, viable cells prior to cryopreservation.
Testing Procedures Include:
- Communicable disease testing performed by a CLIA certified lab.
- Before the release and distribution of any product, our lots are quarantined for 14 days and thoroughly tested for bacterial contamination.
- Plate testing is conducted in multiple phases of the production process to ensure there is zero contamination.
Diseases that are tested prior to donation eligibility:
- Hepatitis Bs Ag
- Hepatitis Bc Ab
- HTLV I/II Ab
- Hepatitis C Ab
- HIV 1&2 Plus O Ab
- CMV ab
- RPR (Non-treponemal syphilis)
- HIV-1/HCV/HBV NAT (Ultrio)
- WNV NAT
Processing this proprietary process meets the FDA’s requirements being minimally manipulated, include:
- For structural tissue, processing that does not alter the original relevant characteristics of the tissue related to the tissue’s utility for reconstruction, repair or replacement; and
- For cells or nonstructural tissues, processing that does not alter the relevant biological characteristics of cells or tissues.
- Transfer means the placement of human reproductive cells or tissues into a human recipient.
Cryopreservation: We use a unique cryopreservation technique that includes using the donation’s own natural components integrated with a natural cryoprotectant to help ensure maximum functional cells post-thaw for homologous use.
Shipping: Our products are shipped overnight in cryopreserved authorized shipping containers. Products are shipped only to licensed medical professionals.
